Propoxyphene 224 was produced when propionic anhydride is added to compound 223 and heated to reflux122 (Fig. 51). Among the safest and most efficient antibiotics for the human body system, it is one of the medications on the WHO essential pharmaceuticals list.95 Doxycycline is an isomer of tetracycline with one hydroxyl group different from the other. Chemically it is also known as (4S,4aR,5S,5aR,6R,12aS)-4-(dimethylamino)-3,5,10,12,12a-pentahydroxy-6-methyl-1,11-dioxo-1,4,4a,5,5a,6,11,12a-octahydrotetracene-2-carboxamide. The oxidation step of the homoallyl system, with the exception that N-chlorosuccinimide is employed as the oxidant.
Fig 18 Synthesis Of Sumatriptan
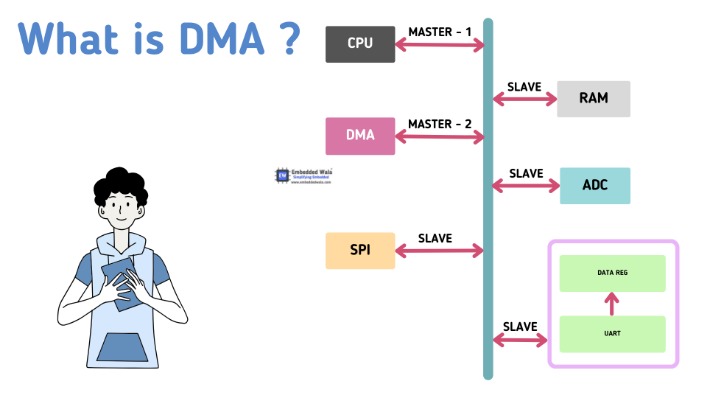
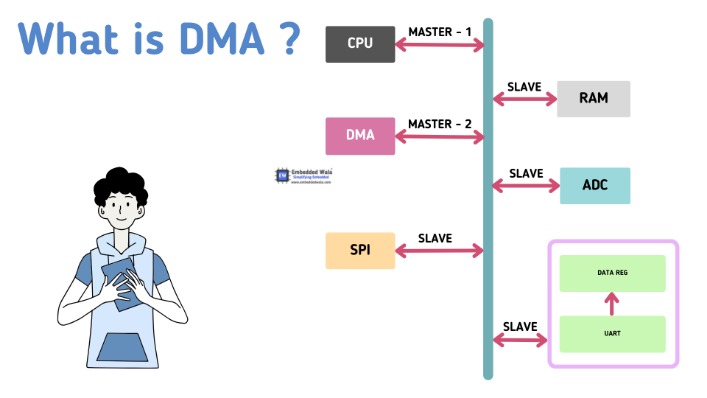
Common synthetic routes involve the modification of the DMA scaffold through functional group manipulation, scaffold hopping, or combinatorial chemistry approaches. Therapeutically, DMA derivatives have shown promise in the treatment of infectious diseases, especially bacterial infections. Additionally, by focusing on particular biochemical pathways involved in tumor growth and metastasis, DMA-based drugs have shown anticancer activity. In addition to their direct pharmacological effects, DMA derivatives can serve as valuable tools in drug delivery systems, prodrug design, and molecular imaging techniques, enhancing their utility in medicinal chemistry research.
Fig 37 Synthesis Of Pyrilamine
When pheniramine is taken in lethal doses, it can cause cardiac arrhythmias, dilated pupils, urine retention, tachycardia, dry flushed skin, decreased bowel noises, confusion, and a little rise in body temperature. The majority of the side effects occurs due to its antimuscarinic action.66 It is used in the treatment of allergies, hay fever, and common cold. The chemical name of pheniramine is N,N-dimethyl-3-phenyl-3-(pyridin-2-yl)propan-1-amine. Trimipramine, also known as surmontil, is an antidepressant that also acts as a sedative to lessen anxiety.
Fig 47 Synthesis Of Methscopolamine Bromide
Using one of the two methods for DMA and the method for nitrite on a given sample can show the possibility of NDMA formation. This does not suggest the sample has NDMA or will develop NDMA, but does suggest a potential for NDMA formation. For example, the 500 mg metformin tablet contains 42.6 μg/g DMA and 6.86 μg/g nitrite.
Fig 40 Structure Of Chlortetracycline Hydrochloride
For many years, the synthesis for this compound, developed by Novartis, used tributyltin azide to form the tetrazole, with xylene as a solvent. However, in 2014, China’s Zhejiang Huahai Pharmaceutical, which makes valsartan for some companies, filed a patent for an improved method for forming the tetrazole ring. The new route involved swapping out tributyltin azide for sodium azide, which leads to higher yields. So all you would need in order to form NDMA in this situation is some form of a nitrosating agent, Light says.

Compound 87 was finally condensed using 3-(dimethylamino) propyl chloride (compound 88) utilizing DMSO and NaH, to yield citalopram 87 (ref. 57) (Fig. 23). Sumatriptan is a selective serotonin agonist, used in the management of migraine. Chemically it is known as 1-(3-(2-(dimethylamino)ethyl)-1H-indol-5-yl)-N-methylmethanesulfonamide. It is synthesized by reacting ethyl (((3-amino-1H-indol-5-yl)methyl)sulfonyl)(methyl)carbamate 58 with NaNO2 in the presence of HCl to form diazonium salt 59, which reacts with ethyl 2-acetyl-5-(dimethylamino)pentanoate 60 to give compound 61. The reaction of compound 62 with MeOH in the presence of KOH resulted in compound 63. The compound 63 on decarboxylation in the presence of copper in quinolone at 200 °C to form sumatriptan 64 (ref. 48 and 49) (Fig. 18).

If someone does not address serotonin syndrome, it can cause life threatening complications, including seizures, kidney or respiratory failure, or loss of muscle tissue. The experience can be so powerful that users may have difficulty processing and integrating the “trip” into real life. Because DMT can mimic a near-death experience, some people may find using the drug traumatic and upsetting.
Fig 33 Synthesis Of Promethazine Hydrochloride
Additionally, incorporating antioxidants like ascorbic acid and alpha-tocopherol can inhibit the nitrosation reaction. This article provides a comprehensive overview that can aid researchers in development of innovative DMA-based drugs, ensuring continued progress in the field of pharmaceuticals. There are various methods to synthesize DMA-containing drugs, such as the Eschweiler–Clarke reaction and the substitution reaction of alkylamine with methyl halide. However, these reactions have several drawbacks, including the use of hazardous chemicals, high waste production, and often poor selectivity. To address these issues, transition metal-catalyzed N-methylation of amines with methyl sources such as formic acid, carbon dioxide, and paraformaldehyde has been employed.
- The chemical name of brompheniramine maleate is 3-(4-bromophenyl)-N,N-dimethyl-3-(pyridin-2-yl)propan-1-amine maleate.
- The latter subunit, if acetylated, binds Brd4, which in turn enhances the transcriptional activation of NF-ĸB and the expression of a subset of NF-ĸB-responsive inflammatory genes.
- Certain prescription and OTC medicines may also give a positive test result.
- 2,5-DMA belongs to the DOx family of amphetamines, which contain methoxy groups on the 2 and 5 positions of the benzene ring, and is also their simplest member.
- Overall, DMA derivatives represent a versatile class of compounds with immense potential in medicinal chemistry.
Fig 59 Synthesis Of Topotecan Hydrochloride

Pyrilamine rapidly permeates the brain and causes drowsiness.87 It was patented in 1943 and used in medicine in 1949. It is sold under the brand names Prefrin-A, Neo-Pyramine, Histadyl, Neo-Antergan, and Nisaval.88 The chemical name of pyrilamine is N1-(4-methoxybenzyl)-N2,N2-dimethyl-N1-(pyridine-2-yl)ethane-1,2-diamine. It is synthesized by reacting 4-methoxy benzaldehyde 154 with pyridine-2-amine 155 in presence of formic acid to form compound 156, which on reacting with 2-chloro-N,N-dimethylethan-1-amine 157 to give pyrilamine 158. On reacting compound 158 with maleic acid target molecule pyrilamine 159 maleate was obtained89 (Fig. 37). Carbinoxamine is a first-generation antihistaminic H1 receptor antagonist. It is used in the treatment of rhinitis, hay fever, urticaria, dermatographism, and allergic conjunctivitis.83 The chemical name of carbinoxamine is 2-((4-chlorophenyl)(pyridine-2-yl)methoxy)-N,N-dimethylethan-1-amine.
Structurally, MDA and MDMA are related compounds with a shared chemical backbone. In fact, MDMA is derived from MDA through a chemical modification process. It would be undoubtedly useful to have a multiresidue method, validated on multiple matrices at the same time, in order to obtain a forensic analysis as more reliable as possible.

Health Encyclopedia
The toxicity and long-term health effects of recreational 2,5-DMA use do not seem to have been studied in any scientific context and the exact toxic dose is unknown. This is because 2,5-DMA is a research chemical with very little history of human usage. It may or may not be more active when taken sublingually.citation needed. However, it is unknown whether this would result in typical psychedelic effects. Unlike its phenethylamine counterpart, 2C-H, it does not appear to be inactive. Clarithromycin is structurally different from erythromycin in that it has an o-methyl substitution at 6th position of the lactone ring, which results in enhanced antibacterial and pharmacokinetic qualities as well as acid stability.
Smoking or food intake can also contaminate the oral fluid, causing an altered response 82. Rinsing the mouth before sample collection can help to reduce any residual contamination 40. For most substances, the concentration in this matrix can be estimated on the basis of oral fluid pH in respect to plasma pH, and the pKa of the drug 71, 72.